Establish Yourself as the Leader in ADC Toxicology Services
With NHP shortages increasing the cost of in vivo studies, ADC drug developers are prioritizing better understanding the toxicity of their drug in vitro to improve asset prioritization and reduce costs. If you’re an in vitro toxicity assay provider join us to establish your expertise in improved prediction and capture this growing market opportunity...
As ADC companies turn to Asian CROs to conduct their toxicology studies with price and reliability forefront of mind, logistical challenges still exist working in Asia. The 3rd ADC Toxicity Summit offers the perfect venture to outline how you offer the perfect balance of cost, reliability, expertise and ease to work with.
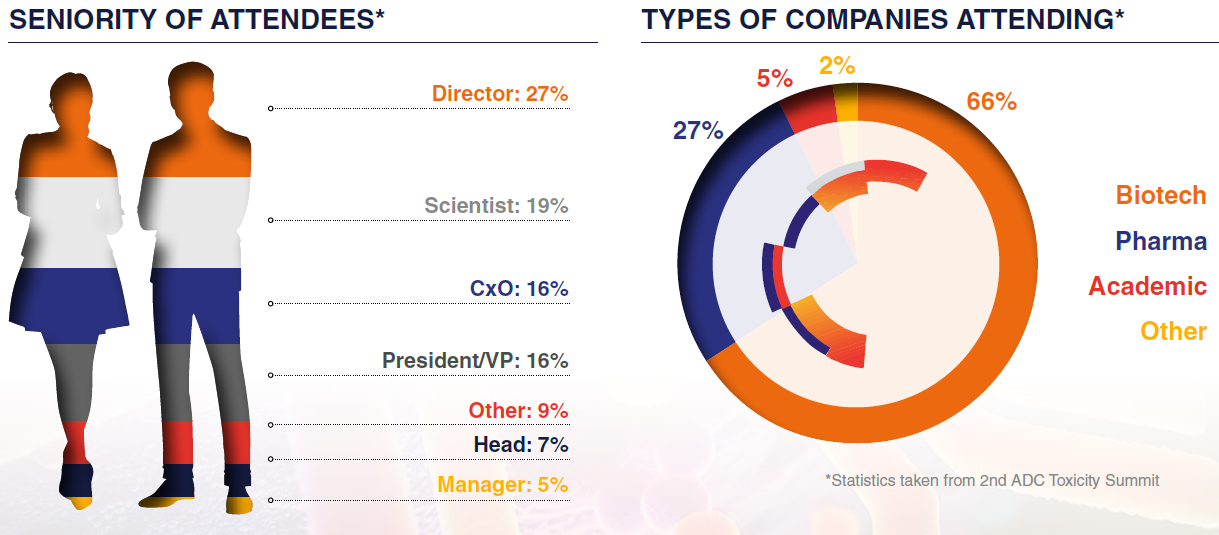
With an audience of toxicologists, pathologists, and preclinical and translational scientists, they are the ones using your services, making this a stand-out opportunity to gain technical and business buy-in to your company.
Whether you're looking to align your brand, showcase your technical expertise, or connect with decision-makers, we offer tailored sponsorship opportunities to help you meet your business goals.
Partnership Options Include: